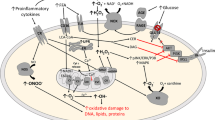
Abstract
Although assays for the most popular markers of exercise-induced oxidative stress may experience methodological flaws, there is sufficient credible evidence to suggest that exercise is accompanied by an increased generation of free radicals, resulting in a measurable degree of oxidative modifications to various molecules. However, the mechanisms responsible are unclear. A common assumption that increased mitochondrial oxygen consumption leads per se to increased reactive oxygen species (ROS) production is not supported by in vitro and in vivo data. The specific contributions of other systems (xanthine oxidase, inflammation, haem protein auto-oxidation) are poorly characterised. It has been demonstrated that ROS have the capacity to contribute to the development of muscle fatigue in situ, but there is still a lack of convincing direct evidence that ROS impair exercise performance in vivo in humans. It remains unclear whether exercise-induced oxidative modifications have little significance, induce harmful oxidative damage, or are an integral part of redox regulation. It is clear that ROS play important roles in numerous physiological processes at rest; however, the detailed physiological functions of ROS in exercise remain to be elucidated.






Similar content being viewed by others
References
Knight JA. Review: free radicals, antioxidants, and the immune system. Ann Clin Lab Sci 2000; 30 (2): 145–158
Turpaev KT. Reactive oxygen species and regulation of gene expression. Biochemistry (Mosc) 2002; 67 (3): 281–292
Thannickal VJ, Fanburg BL. Reactive oxygen species in cell signaling. Am J Physiol Lung Cell Mol Physiol 2000; 279 (6): L1005–L1028
Cooper CE. Ferryl states and protein free radicals. In: Rice-Evans CA, Burdon RH, editors. Free radical damage and its control. Amsterdam: Elsevier, 1993: 67–111
Halliwell B, Chirico S. Lipid peroxidation: its mechanism, measurement, and significance. Am J Clin Nutr 1993; 57 (5 Suppl.): 715S–724S
Golden TR, Hinerfeld DA, Melov S. Oxidative stress and aging: beyond correlation. Aging Cell 2002; 1 (2): 117–123
Dillard CJ, Litov RE, Savin WM, et al. Effects of exercise, vitamin E, and ozone on pulmonary function and lipid peroxidation. J Appl Physiol 1978; 45 (6): 927–932
Davies KJ, Quintanilha AT, Brooks GA, et al. Free radicals and tissue damage produced by exercise. Biochem Biophys Res Commun 1982; 107 (4): 1198–1205
Ashton T, Rowlands CC, Jones E, et al. Electron spin resonance spectroscopic detection of oxygen-centred radicals in human serum following exhaustive exercise. Eur J Appl Physiol Occup Physiol 1998; 77 (6): 498–502
Ashton T, Young IS, Peters JR, et al. Electron spin resonance spectroscopy, exercise, and oxidative stress: an ascorbic acid intervention study. J Appl Physiol 1999; 87 (6): 2032–2036
Bailey DM, Davies B, Young IS, et al. EPR spectroscopic detection of free radical outflow from an isolated muscle bed in exercising humans. J Appl Physiol 2003; 94 (5): 1714–1718
Bailey DM, Young IS, McEneny J, et al. Regulation of free radical outflow from an isolated muscle bed in exercising humans. Am J Physiol Heart Circ Physiol 2004; 287 (4): H1689–H1699
Groussard C, Rannou-Bekono F, Machefer G, et al. Changes in blood lipid peroxidation markers and antioxidants after a single sprint anaerobic exercise. Eur J Appl Physiol 2003; 89 (1): 14–20
Viinikka L, Vuori J, Ylikorkala O. Lipid peroxides, prostacyclin, and thromboxane A2 in runners during acute exercise. Med Sci Sports Exerc 1984; 16 (3): 275–277
Maughan RJ, Donnelly AE, Gleeson M, et al. Delayed-onset muscle damage and lipid peroxidation in man after a downhill run. Muscle Nerve 1989; 12 (4): 332–336
Duthie GG, Robertson JD, Maughan RJ, et al. Blood antioxidant status and erythrocyte lipid peroxidation following distance running. Arch Biochem Biophys 1990; 282 (1): 78–83
Sahlin K, Ekberg K, Cizinsky S. Changes in plasma hypoxanthine and free radical markers during exercise in man. Acta Physiol Scand 1991; 142 (2): 275–281
Hartmann A, Niess AM, Grunert-Fuchs M, et al. Vitamin E prevents exercise-induced DNA damage. Mutat Res 1995; 346 (4): 195–202
Vasankari T, Kujala U, Heinonen O, et al. Measurement of serum lipid peroxidation during exercise using three different methods: diene conjugation, thiobarbituric acid reactive material and fluorescent chromolipids. Clin Chim Acta 1995; 234 (1–2): 63–69
Niess AM, Hartmann A, Grunert-Fuchs M, et al. DNA damage after exhaustive treadmill running in trained and untrained men. Int J Sports Med 1996; 17 (6): 397–403
Dufaux B, Heine O, Kothe A, et al. Blood glutathione status following distance running. Int J Sports Med 1997; 18 (2): 89–93
Margaritis I, Tessier F, Richard MJ, et al. No evidence of oxidative stress after a triathlon race in highly trained competitors. Int J Sports Med 1997; 18 (3): 186–190
Chung SC, Goldfarb AH, Jamurtas AZ, et al. Effect of exercise during the follicular and luteal phases on indices of oxidative stress in healthy women. Med Sci Sports Exerc 1999; 31 (3): 409–413
Leaf DA, Kleinman MT, Hamilton M, et al. The effect of exercise intensity on lipid peroxidation. Med Sci Sports Exerc 1997; 29 (8): 1036–1039
Surmen-Gur E, Ozturk E, Gur H, et al. Effect of vitamin E supplementation on post-exercise plasma lipid peroxidation and blood antioxidant status in smokers: with special reference to haemoconcentration effect. Eur J Appl Physiol Occup Physiol 1999; 79 (6): 472–478
Alessio HM, Hagerman AE, Fulkerson BK, et al. Generation of reactive oxygen species after exhaustive aerobic and isometric exercise. Med Sci Sports Exerc 2000; 32 (9): 1576–1581
Sacheck JM, Decker EA, Clarkson PM. The effect of diet on vitamin E intake and oxidative stress in response to acute exercise in female athletes. Eur J Appl Physiol 2000; 83 (1): 40–46
Akova B, Surmen-Gur E, Gur H, et al. Exercise-induced oxidative stress and muscle performance in healthy women: role of vitamin E supplementation and endogenous oestradiol. Eur J Appl Physiol 2001; 84 (1–2): 141–147
Quindry JC, Stone WL, King J, et al. The effects of acute exercise on neutrophils and plasma oxidative stress. Med Sci Sports Exerc 2003; 35 (7): 1139–1145
Sacheck JM, Milbury PE, Cannon JG, et al. Effect of vitamin E and eccentric exercise on selected biomarkers of oxidative stress in young and elderly men. Free Radic Biol Med 2003; 34 (12): 1575–1588
Ramel A, Wagner KH, Elmadfa I. Plasma antioxidants and lipid oxidation after submaximal resistance exercise in men. Eur J Nutr 2004; 43 (1): 2–6
Lovlin R, Cottle W, Pyke I, et al. Are indices of free radical damage related to exercise intensity? Eur J Appl Physiol Occup Physiol 1987; 56 (3): 313–316
Kretzschmar M, Muller D, Hubscher J, et al. Influence of aging, training and acute physical exercise on plasma glutathione and lipid peroxides in man. Int J Sports Med 1991; 12 (2): 218–222
Bird RP, Draper HH. Comparative studies on different methods of malonaldehyde determination. Methods Enzymol 1984; 105: 299–305
Lepage G, Munoz G, Champagne J, et al. Preparative steps necessary for the accurate measurement of malondialdehyde by high-performance liquid chromatography. Anal Biochem 1991; 197 (2): 277–283
Halliwell B, Gutteridge JMC. Free radicals in biology and medicine. Oxford: University Press, 1998
Kanter MM, Lesmes GR, Kaminsky LA, et al. Serum creatine kinase and lactate dehydrogenase changes following an eighty kilometer race: relationship to lipid peroxidation. Eur J Appl Physiol Occup Physiol 1988; 57 (1): 60–63
Kanter MM, Eddy DE. Effects of antioxidants supplementation on serum markers of lipid peroxidation and skeletal muscle damage following eccentric exercise [abstract]. Med Sci Sports Exerc 1992; 24 (5): S17
Rokitzki L, Logemann E, Sagredos AN, et al. Lipid peroxidation and antioxidative vitamins under extreme endurance stress. Acta Physiol Scand 1994; 151 (2): 149–158
Marzatico F, Pansarasa O, Bertorelli L, et al. Blood free radical antioxidant enzymes and lipid peroxides following long-distance and lactacidemic performances in highly trained aerobic and sprint athletes. J Sports Med Phys Fitness 1997; 37 (4): 235–239
Hessel E, Haberland A, Muller M, et al. Oxygen radical generation of neutrophils: a reason for oxidative stress during marathon running? Clin Chim Acta 2000; 298 (1–2): 145–156
Ji LL. Antioxidant enzyme response to exercise and aging. Med Sci Sports Exerc 1993; 25 (2): 225–231
Largilliere C, Melancon SB. Free malondialdehyde determination in human plasma by high-performance liquid chromatography. Anal Biochem 1988; 170 (1): 123–126
Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol 1978; 52: 302–310
Esterbauer H, Cheeseman KH. Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal. Methods Enzymol 1990; 186: 407–421
Mihara M, Uchiyama M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 1978; 86 (1): 271–278
Carbonneau MA, Peuchant E, Sess D, et al. Free and bound malondialdehyde measured as thiobarbituric acid adduct by HPLC in serum and plasma. Clin Chem 1991; 37 (8): 1423–1429
Hong YL, Yeh SL, Chang CY, et al. Total plasma malondialdehyde levels in 16 Taiwanese college students determined by various thiobarbituric acid tests and an improved high-performance liquid chromatography-based method. Clin Biochem 2000; 33 (8): 619–625
Maxwell SR, Jakeman P, Thomason H, et al. Changes in plasma antioxidant status during eccentric exercise and the effect of vitamin supplementation. Free Radic Res Commun 1993; 19 (3): 191–202
Sen CK, Rankinen T, Vaisanen S, et al. Oxidative stress after human exercise: effect of N-acetylcysteine supplementation. J Appl Physiol 1994; 76 (6): 2570–2577
Vasankari TJ, Kujala UM, Vasankari TM, et al. Effects of acute prolonged exercise on-serum and LDL oxidation and antioxidant defences. Free Radic Biol Med 1997; 22 (3): 509–513
Alessio HM, Goldfarb AH, Cao G. Exercise-induced oxidative stress before and after vitamin C supplementation. Int J Sport Nutr 1997; 7 (1): 1–9
Child RB, Wilkinson DM, Fallowfield JL, et al. Elevated serum antioxidant capacity and plasma malondialdehyde concentration in response to a simulated half-marathon run. Med Sci Sports Exerc 1998; 30 (11): 1603–1607
Szczesniak L, Karolkiewicz J, Deskur E, et al. The influence of exercise-induced oxidative stress on binding and degradation of 125I-insulin by the receptors on erythrocytes. J Physiol Pharmacol 1998; 49 (3): 421–432
Borsheim E, Knardahl S, Hostmark AT. Short-term effects of exercise on plasma very low density lipoproteins (VLDL) and fatty acids. Med Sci Sports Exerc 1999; 31 (4): 522–530
Laaksonen DE, Atalay M, Niskanen L, et al. Blood glutathione homeostasis as a determinant of resting and exercise-induced oxidative stress in young men. Redox Rep 1999; 4 (1–2): 53–59
Leaf DA, Kleinman MT, Hamilton M, et al. The exercise-induced oxidative stress paradox: the effects of physical exercise training. Am J Med Sci 1999; 317 (5): 295–300
Child RB, Wilkinson DM, Fallowfield JL. Effects of a training taper on tissue damage indices, serum antioxidant capacity and half-marathon running performance. Int J Sports Med 2000; 21 (5): 325–331
Roberts LJ, Morrow JD. Products of the isoprostane pathway: unique bioactive compounds and markers of lipid peroxidation. Cell Mol Life Sci 2002; 59 (5): 808–820
Fam SS, Morrow JD. The isoprostanes: unique products of arachidonic acid oxidation — a review. Curr Med Chem 2003; 10 (17): 1723–1740
Waring WS, Convery A, Mishra V, et al. Uric acid reduces exercise-induced oxidative stress in healthy adults. Clin Sci (Lond) 2003; 105 (4): 425–430
McAnulty SR, McAnulty LS, Nieman DC, et al. Influence of carbohydrate ingestion on oxidative stress and plasma antioxidant potential following a 3h run. Free Radic Res 2003; 37 (8): 835–840
Mastaloudis A, Leonard SW, Traber MG. Oxidative stress in athletes during extreme endurance exercise. Free Radic Biol Med 2001; 31 (7): 911–922
Mastaloudis A, Morrow JD, Hopkins DW, et al. Antioxidant supplementation prevents exercise-induced lipid peroxidation, but not inflammation, in ultramarathon runners. Free Radic Biol Med 2004; 36 (10): 1329–1341
Nieman DC, Henson DA, McAnulty SR, et al. Influence of vitamin C supplementation on oxidative and immune changes after an ultramarathon. J Appl Physiol 2002; 92 (5): 1970–1977
Steensberg A, Morrow J, Toft AD, et al. Prolonged exercise, lymphocyte apoptosis and F2-isoprostanes. Eur J Appl Physiol 2002; 87 (1): 38–42
Bailey DM, Davies B, Young IS. Intermittent hypoxic training: Implications for lipid peroxidation induced by acute normoxic exercise in active men. Clin Sci (Lond) 2001; 101 (5): 465–475
Pincemail J, Camus G, Roesgen A, et al. Exercise induces pentane production and neutrophil activation in humans: effect of propranolol. Eur J Appl Physiol Occup Physiol 1990; 61 (3–4): 319–322
Kanter MM, Nolte LA, Holloszy JO. Effects of an antioxidant vitamin mixture on lipid peroxidation at rest and postexercise. J Appl Physiol 1993; 74 (2): 965–969
Liu ML, Bergholm R, Bergholm R, et al. A marathon run increases the susceptibility of LDL to oxidation in vitro and modifies plasma antioxidants. Am J Physiol 1999; 276: E1083–E1091
Case D, Baer JT, Subbiah MT. The effect of prolonged exercise on lipid peroxidation in eumenorrheic female runners. Med Sci Sports Exerc 2004; 31 (10): 1390–1393
Ginsburg GS, Agil A, O’Toole M, et al. Effects of a single bout of ultraendurance exercise on lipid levels and susceptibility of lipids to peroxidation in triathletes. JAMA 1996; 276 (3): 221–225
Viguie CA, Frei B, Shigenaga MK, et al. Antioxidant status and indexes of oxidative stress during consecutive days of exercise. J Appl Physiol 1993; 75 (2): 566–572
Hartmann A, Pfuhler S, Dennog C, et al. Exercise-induced DNA effects in human leukocytes are not accompanied by increased formation of 8-hydroxy-2’-deoxyguanosine or induction of micronuclei. Free Radic Biol Med 1998; 24 (2): 245–251
Nielsen HB, Hanel B, Loft S, et al. Restricted pulmonary diffusion capacity after exercise is not an ARDS-like injury. J Sports Sci 1995; 13 (2): 109–113
Witt EH, Reznick AZ, Viguie CA, et al. Exercise, oxidative damage and effects of antioxidant manipulation. J Nutr 1992; 122 (3 Suppl.): 766–773
Mastaloudis A, Yu TW, O’Donnell RP, et al. Endurance exercise results in DNA damage as detected by the comet assay. Free Radic Biol Med 2004; 36 (8): 966–975
Hartmann A, Plappert U, Raddatz K, et al. Does physical activity induce DNA damage? Mutagenesis 1994; 9 (3): 269–272
Radak Z, Ogonovszky H, Dubecz J, et al. Super-marathon race increases serum and urinary nitrotyrosine and carbonyl levels. Eur J Clin Invest 2003; 33 (8): 726–730
Chevion S, Moran DS, Heled Y, et al. Plasma antioxidant status and cell injury after severe physical exercise. Proc Natl Acad Sci U S A 2003; 100 (9): 5119–5123
Gohil K, Viguie C, Stanley WC, et al. Blood glutathione oxidation during human exercise. J Appl Physiol 1988; 64 (1): 115–119
Camus G, Felekidis A, Pincemail J, et al. Blood levels of reduced/oxidized glutathione and plasma concentration of ascorbic acid during eccentric and concentric exercises of similar energy cost. Arch Int Physiol Biochim Biophys 1994; 102 (1): 67–70
Laires MJ, Madeira F, Sergio J, et al. Preliminary study of the relationship between plasma and erythrocyte magnesium variations and some circulating pro-oxidant and antioxidant indices in a standardized physical effort. Magnes Res 1993; 6 (3): 233–238
Lee J, Goldfarb AH, Rescino MH, et al. Eccentric exercise effect on blood oxidative-stress markers and delayed onset of muscle soreness. Med Sci Sports Exerc 2002; 34 (3): 443–448
Rossi R, Milzani A, Dalle-Donne I, et al. Blood glutathione disulfide: in vivo factor or in vitro artifact? Clin Chem 2002; 48 (5): 742–753
Sastre J, Asensi M, Gasco E, et al. Exhaustive physical exercise causes oxidation of glutathione status in blood: prevention by antioxidant administration. Am J Physiol 1992; 263 (5 Pt 2): R992–R995
Vina J, Sastre J, Asensi M, et al. Assay of blood glutathione oxidation during physical exercise. Methods Enzymol 1995; 251: 237–243
Pincemail J, Deby C, Camus G, et al. Tocopherol mobilization during intensive exercise. Eur J Appl Physiol Occup Physiol 1988; 57 (2): 189–191
Kawai Y, Iwane H, Takanami Y. Vitamin E is mobilized in relation to lipolysis after strenuous endurance exercise [abstract]. Med Sci Sports Exerc 1994; 26 (5): S7
Kawai Y, Iwane H, Takanami Y. Mechanisms of the increase in serum vitamin E level through strenuous endurance exercise [abstract]. Med Sci Sports Exerc 1995; 27 (5): S9
Camus G, Pincemail J, Roesgen A, et al. Tocopherol mobilization during dynamic exercise after beta-adrenergic blockade. Arch Int Physiol Biochim 1990; 98 (1): 121–126
Kawai Y, Shimomitsu T, Takanami Y, et al. Vitamin E level changes in serum and red blood cells due to acute exhaustive exercise in collegiate women. J Nutr Sci Vitaminol (Tokyo) 2000; 46 (3): 119–124
Schneider M, Niess AM, Rozario F, et al. Vitamin E supplementation does not increase the vitamin C radical concentration at rest and after exhaustive exercise in healthy male subjects. Eur J Nutr 2003; 42 (4): 195–200
Garry PJ, Appenzeller O. Vitamins A and C and endurance races. Ann Sports Med 1983; 1: 82–84
Gleeson M, Robertson JD, Maughan RJ. Influence of exercise on ascorbic acid status in man. Clin Sci (Colch) 1987; 73 (5): 501–505
Inoue T, Mu Z, Sumikawa K, et al. Effect of physical exercise on the content of 8-hydroxydeoxyguanosine in nuclear DNA prepared from human lymphocytes. Jpn J Cancer Res 1993; 84 (7): 720–725
Poulsen HE, Loft S, Vistisen K. Extreme exercise and oxidative DNA modification. J Sports Sci 1996; 14 (4): 343–346
Koyama K, Kaya M, Ishigaki T, et al. Role of xanthine oxidase in delayed lipid peroxidation in rat liver induced by acute exhausting exercise. Eur J Appl Physiol Occup Physiol 1999; 80 (1): 28–33
Liu J, Yeo HC, Overvik-Douki E, et al. Chronically and acutely exercised rats: biomarkers of oxidative stress and endogenous antioxidants. J Appl Physiol 2000; 89 (1): 21–28
Boveris A, Oshino N, Chance B. The cellular production of hydrogen peroxide. Biochem J 1972; 128 (3): 617–630
Boveris A, Chance B. The mitochondrial generation of hydrogen peroxide: general properties and effect of hyperbaric oxygen. Biochem J 1973; 134 (3): 707–716
Herrero A, Barja G. ADP-regulation of mitochondrial free radical production is different with complex I- or complex II-linked substrates: implications for the exercise paradox and brain hypermetabolism. J Bioenerg Biomembr 1997; 29 (3): 241–249
Murphy MP, Echtay KS, Blaikie FH, et al. Superoxide activates uncoupling proteins by generating carbon-centered radicals and initiating lipid peroxidation: studies using a mitochondria-targeted spin trap derived from alpha-phenyl-N-tert-butylnitrone. J Biol Chem 2003; 278 (49): 48534–48545
Richardson RS, Leigh JS, Wagner PD, et al. Cellular pO2 as a determinant of maximal mitochondrial (2) consumption in trained human skeletal muscle. J Appl Physiol 1999; 87 (1): 325–331
Richardson RS, Newcomer SC, Noyszewski EA. Skeletal muscle intracellular P (2) assessed by myoglobin desaturation: response to graded exercise. J Appl Physiol 2001; 91 (6): 2679–2685
Bailey DM. What regulates exercise-induced reactive oxidant generation: mitochondrial (2) flux or PO(2)? Med Sci Sports Exerc 2001; 33 (4): 681–682
Drahota Z, Chowdhury SK, Floryk D, et al. Glycerophosphate-dependent hydrogen peroxide production by brown adipose tissue mitochondria and its activation by ferricyanide. J Bioenerg Biomembr 2002; 34: 105–113
Miwa S, St-Pierre J, Partridge L, et al. Superoxide and hydrogen peroxide production by Drosophila mitochondria. Free Rad Biol Med 2003; 35 (8): 938–948
MacDonald MJ, Brown LJ. Calcium activation of mitochondrial glycerol phosphate dehydrogenase restudied. Arch Biochem Biophys 1996; 326 (1): 79–84
Rasmussen UF, Krustrup P, Bangsbo J, et al. The effect of high-intensity exhaustive exercise studied in isolated mitochondria from human skeletal muscle. Pflugers Arch 2001; 443 (2): 180–187
Jesina P, Kholova D, Bolehovska R, et al. Glycerophosphate-dependent hydrogen peroxide production by rat liver mitochondria. Physiol Res 2004; 53 (3): 305–310
Packer L. Oxidants, antioxidant nutrients and the athlete. J Sports Sci 1997; 15 (3): 353–363
Roy RS, McCord JM. Superoxide and ischemia: conversion of xanthine dehydrogenase to xanthine oxidase. In: Greenvald R, Cohen G, editors. Oxy radicals and their scavenger systems. New York: Elsevier Science Publishing, 1983: 145–153
Volek JS, Kraemer WJ, Rubin MR, et al. L-carnitine L-tartrate supplementation favorably affects markers of recovery from exercise stress. Am J Physiol Endocrinol Metab 2002; 282 (2): E474–E482
Hellsten Y, Hansson HA, Johnson L, et al. Increased expression of xanthine oxidase and insulin-like growth factor I (IGF-I) immunoreactivity in skeletal muscle after strenuous exercise in humans. Acta Physiol Scand 1996; 157 (2): 191–197
Hellsten Y, Frandsen U, Orthenblad N, et al. Xanthine oxidase in human skeletal muscle following eccentric exercise: a role in inflammation. J Physiol 1997; 498 (Pt 1): 239–248
Xia Y, Zweier JL. Substrate control of free radical generation from xanthine oxidase in the postischemic heart. J Biol Chem 1995; 270 (32): 18797–18803
Bangsbo J, Sjodin B, Hellsten-Westing Y. Exchange of hypoxanthine in muscle during intense exercise in man. Acta Physiol Scand 1992; 146 (4): 549–550
Hellsten-Westing Y, Kaijser L, Ekblom B, et al. Exchange of purines in human liver and skeletal muscle with short-term exhaustive exercise. Am J Physiol 1994; 266 (1 Pt 2): R81–R86
Hellsten Y, Ahlborg G, Jensen-Urstad M, et al. Indication of in vivo xanthine oxidase activity in human skeletal muscle during exercise. Acta Physiol Scand 1988; 134 (1): 159–160
Vina J, Gimeno A, Sastre J, et al. Mechanism of free radical production in exhaustive exercise in humans and rats; role of xanthine oxidase and protection by allopurinol. IUBMB Life 2000; 49 (6): 539–544
Heunks LM, Vina J, van Herwaarden CL, et al. Xanthine oxidase is involved in exercise-induced oxidative stress in chronic obstructive pulmonary disease. Am J Physiol 1999; 277 (6 Pt 2): R1697–R1704
Pyne DB. Exercise-induced muscle damage and inflammation: a review. Aust J Sci Med Sport 1994; 26 (3–4): 49–58
Morozov VI, Pryatkin SA, Kalinski MI, et al. Effect of exercise to exhaustion on myeloperoxidase and lysozyme release from blood neutrophils. Eur J Appl Physiol 2003; 89 (3–4): 257–262
Yamada M, Suzuki K, Kudo S, et al. Effect of exhaustive exercise on human neutrophils in athletes. Luminescence 2000; 15 (1): 15–20
Suzuki K, Nakaji S, Yamada M, et al. Impact of a competitive marathon race on systemic cytokine and neutrophil responses. Med Sci Sports Exerc 2003; 35 (2): 348–355
Suzuki K, Sato H, Kikuchi T, et al. Capacity of circulating neutrophils to produce reactive oxygen species after exhaustive exercise. J Appl Physiol 1996; 81 (3): 1213–1222
Bury TB, Pirnay F. Effect of prolonged exercise on neutrophil myeloperoxidase secretion. Int J Sports Med 1995; 16 (6): 410–412
Camus G, Pincemail J, Ledent M, et al. Plasma levels of polymorphonuclear elastase and myeloperoxidase after uphill walking and downhill running at similar energy cost. Int J Sports Med 1992; 13 (6): 443–446
Wetzstein CJ, Shern-Brewer RA, Santanam N, et al. Does acute exercise affect the susceptibility of low density lipoprotein to oxidation? Free Radic Biol Med 1998; 24 (4): 679–682
Niess AM, Sommer M, Schlotz E, et al. Expression of the inducible nitric oxide synthase (iNOS) in human leukocytes: responses to running exercise. Med Sci Sports Exerc 2000; 32 (7): 1220–1225
Childs A, Jacobs C, Kaminski T, et al. Supplementation with vitamin C and N-acetyl-cysteine increases oxidative stress in humans after an acute muscle injury induced by eccentric exercise. Free Radic Biol Med 2001; 31 (6): 745–753
Cooper CE, Vollaard NB, Choueiri T, et al. Exercise, free radicals and oxidative stress. Biochem Soc Trans 2002; 30 (2): 280–284
Emerit J, Beaumont C, Trivin F. Iron metabolism, free radicals, and oxidative injury. Biomed Pharmacother 2001; 55 (6): 333–339
Gibson JF, Ingram DJ, Perutz MF. Orientation of the four haem groups in haemoglobin. Nature 1956; 178 (4539): 906–908
Svistunenko DA, Patel RP, Voloshchenko SV, et al. The globin-based free radical of ferryl hemoglobin is detected in normal human blood. J Biol Chem 1997; 272 (11): 7114–7121
Balagopalakrishna C, Manoharan PT, Abugo OO, et al. Production of superoxide from hemoglobin-bound oxygen under hypoxic conditions. Biochemistry 1996; 35 (20): 6393–6398
Vollaard NBJ, Reeder BJ, Shearman JP, et al. A new sensitive assay reveals that hemoglobin is oxidatively modified in vivo. Free Radic Biol Med 2005; 39 (9): 1216–1228
Kanter M. Free radicals, exercise and antioxidant supplementation. Proc Nutr Soc 1998; 57 (1): 9–13
Elosua R, Molina L, Fito M, et al. Response of oxidative stress biomarkers to a 16-week aerobic physical activity program, and to acute physical activity, in healthy young men and women. Atherosclerosis 2003; 167 (2): 327–334
Criswell D, Powers S, Dodd S, et al. High intensity training-induced changes in skeletal muscle antioxidant enzyme activity. Med Sci Sports Exerc 1993; 25 (10): 1135–1140
Higuchi M, Cartier LJ, Chen M, et al. Superoxide dismutase and catalase in skeletal muscle: adaptive response to exercise. J Gerontol 1985; 40 (3): 281–286
Leeuwenburgh C, Fiebig R, Chandwaney R, et al. Aging and exercise training in skeletal muscle: responses of glutathione and antioxidant enzyme systems. Am J Physiol 1994; 267 (2 Pt 2): R439–R445
Powers SK, Criswell D, Lawler J, et al. Influence of exercise and fiber type on antioxidant enzyme activity in rat skeletal muscle. Am J Physiol 1994; 266 (2 Pt 2): R375–R380
Powers SK, Lawler J, Criswell D, et al. Aging and respiratory muscle metabolic plasticity: effects of endurance training. J Appl Physiol 1992; 72 (3): 1068–1073
Oh-ishi S, Kizaki T, Nagasawa J, et al. Effects of endurance training on superoxide dismutase activity, content and mRNA expression in rat muscle. Clin Exp Pharmacol Physiol 1997; 24 (5): 326–332
Oh-ishi S, Kizaki T, Ookawara T, et al. Endurance training improves the resistance of rat diaphragm to exercise-induced oxidative stress. Am J Respir Crit Care Med 1997; 156 (5): 1579–1585
Somani SM, Husain K. Exercise training alters kinetics of antioxidant enzymes in rat tissues. Biochem Mol Biol Int 1996; 38 (3): 587–595
Halliwell B. Free radicals, proteins and DNA: oxidative damage versus redox regulation. Biochem Soc Trans 1996; 24 (4): 1023–1027
Evelson P, Travacio M, Repetto M, et al. Evaluation of total reactive antioxidant potential (TRAP) of tissue homogenates and their cytosols. Arch Biochem Biophys 2001; 388 (2): 261–266
Di Meo S, Venditti P, De Leo T. Tissue protection against oxidative stress. Experientia 1996; 52 (8): 786–794
Cao G, Giovanoni M, Prior RL. Antioxidant capacity in different tissues of young and old rats. Proc Soc Exp Biol Med 1996; 211 (4): 359–365
Xu KY, Zweier JL, Becker LC. Hydroxyl radical inhibits sarcoplasmic reticulum C (2+)-ATPase function by direct attack on the ATP binding site. Circ Res 1997; 80 (1): 76–81
Morris TE, Sulakhe PV. Sarcoplasmic reticulum C (2+)-pump dysfunction in rat cardiomyocytes briefly exposed to hydroxyl radicals. Free Radic Biol Med 1997; 22 (1-2): 37–47
Scherer NM, Deamer DW. Oxidation of thiols in the Ca2+-ATPase of sarcoplasmic reticulum microsomes. Biochim Biophys Acta 1986; 862 (2): 309–317
Scherer NM, Deamer DW. Oxidative stress impairs the function of sarcoplasmic reticulum by oxidation of sulfhydryl groups in the Ca2+-ATPase. Arch Biochem Biophys 1986; 246 (2): 589–601
Lawler JM, Hu Z, Barnes WS. Effect of reactive oxygen species on K+ contractures in the rat diaphragm. J Appl Physiol 1998; 84 (3): 948–953
Sen CK, Kolosova I, Hanninen O, et al. Inward potassium transport systems in skeletal muscle derived cells are highly sensitive to oxidant exposure. Free Radic Biol Med 1995; 18 (4): 795–800
Haycock JW, Jones P, Harris JB, et al. Differential susceptibility of human skeletal muscle proteins to free radical induced oxidative damage: a histochemical, immunocytochemical and electron microscopical study in vitro. Acta Neuropathol (Berl) 1996; 92 (4): 331–340
Lamb GD, Posterino GS. Effects of oxidation and reduction on contractile function in skeletal muscle fibres of the rat. J Physiol 2003; 546 (Pt 1): 149–163
Oba T, Koshita M, Yamaguchi M. H2O2 modulates twitch tension and increases Po of Ca2+ release channel in frog skeletal muscle. Am J Physiol 1996; 271 (3 Pt 1): C810–C818
Reid MB, Khawli FA, Moody MR. Reactive oxygen in skeletal muscle III: contractility of unfatigued muscle. J Appl Physiol 1993; 75 (3): 1081–1087
Barclay JK, Hansel M. Free radicals may contribute to oxidative skeletal muscle fatigue. Can J Physiol Pharmacol 1991; 69 (2): 279–284
Andrade FH, Reid MB, Allen DG, et al. Effect of hydrogen peroxide and dithiothreitol on contractile function of single skeletal muscle fibres from the mouse. J Physiol 1998; 509: 565–575
Reid MB. Invited review: redox modulation of skeletal muscle contraction: what we know and what we don’t. J Appl Physiol 2001; 90 (2): 724–731
Novelli GP, Bracciotti G, Falsini S. Spin-trappers and vitamin E prolong endurance to muscle fatigue in mice. Free Radic Biol Med 1990; 8 (1): 9–13
Novelli GP, Falsini S, Bracciotti G. Exogenous glutathione increases endurance to muscle effort in mice. Pharmacol Res 1991; 23 (2): 149–155
Leeuwenburgh C, Ji LL. Glutathone and glutathione ethyl ester supplementation of mice alter glutathione homeostasis during exercise. J Nutr 1998; 128 (12): 2420–2426
Travaline MJ, Sudarshan S, Roy BG, et al. Effect of N-acetylcysteine on human diaphragm strength and fatigability. Am J Respir Crit Care Med 1997; 156 (5): 1567–1571
Reid MB, Stokic DS, Koch SM, et al. N-acetylcysteine inhibits muscle fatigue in humans. J Clin Invest 1994; 94 (6): 2468–2474
Medved I, Brown MJ, Bjorksten AR, et al. N-acetylcysteine infusion alters blood redox status but not time to fatigue during intense exercise in humans. J Appl Physiol 2003; 94 (4): 1572–1582
Medved I, Brown MJ, Bjorksten AR, et al. Effects of intravenous N-acetylcysteine infusion on time to fatigue and potassium regulation during prolonged exercise. J Appl Physiol 2003; 96 (1): 211–217
Shephard RJ, Campbell R, Pimm P, et al. Vitamin E, exercise, and the recovery from physical activity. Eur J Appl Physiol Occup Physiol 1974; 33 (2): 119–126
Watt T, Romet TT, McFarlane I, et al. Vitamin E and oxygen consumption [letter]. Lancet 1974; II (7876): 354–355
Lawrence JD, Bower RC, Riehl WP, et al. Effects of alpha-tocopherol acetate on the swimming endurance of trained swimmers. Am J Clin Nutr 1975; 28 (3): 205–208
Sharman IM, Down MG, Norgan NG. The effects of vitamin E on physiological function and athletic performance of trained swimmers. J Sports Med Phys Fitness 1976; 16 (3): 215–225
Braun B, Clarkson PM, Freedson PS, et al. Effects of coenzyme Q10 supplementation on exercise performance, VO2max, and lipid peroxidation in trained cyclists. Int J Sport Nutr 1991; 1 (4): 353–365
Snider IP, Bazzarre TL, Murdoch SD, et al. Effects of coenzyme athletic performance system as an ergogenic aid on endurance performance to exhaustion. Int J Sport Nutr 1992; 2 (3): 272–286
Rokitzki L, Logemann E, Huber G, et al. Alpha-tocopherol supplementation in racing cyclists during extreme endurance training. Int J Sport Nutr 1994; 4 (3): 253–264
Laaksonen R, Fogelholm M, Himberg JJ, et al. Ubiquinone supplementation and exercise capacity in trained young and older men. Eur J Appl Physiol Occup Physiol 1995; 72 (1–2): 95–100
Malm C, Svensson M, Ekblom B, et al. Effects of ubiquinone-10 supplementation and high intensity training on physical performance in humans. Acta Physiol Scand 1997; 161 (3): 379–384
Weston SB, Zhou S, Weatherby RP, et al. Does exogenous coenzyme Q10 affect aerobic capacity in endurance athletes? Int J Sport Nutr 1997; 7 (3): 197–206
Itoh H, Ohkuwa T, Yamazaki Y, et al. Vitamin E supplementation attenuates leakage of enzymes following 6 successive days of running training. Int J Sports Med 2000; 21 (5): 369–374
Patwell DM, Jackson MJ. Contraction-induced oxidants as mediators of adaptation and damage in skeletal muscle. Exerc Sport Sci Rev 2004; 32 (1): 14–18
Jackson MJ. Free radicals in skin and muscle: damaging agents or signals for adaptation? Proc Nutr Soc 1999; 58 (3): 673–676
Iwai K, Drake SK, Wehr NB, et al. Iron-dependent oxidation, ubiquitination, and degradation of iron regulatory protein 2: implications for degradation of oxidized proteins. Proc Natl Acad Sci U S A 1998; 95 (9): 4924–4928
Kang DK, Jeong J, Drake SK, et al. Iron regulatory protein 2 as iron sensor: iron-dependent oxidative modification of cysteine. J Biol Chem 2003; 278 (17): 14857–14864
Zhu H, Bunn HF. Signal transduction: how do cells sense oxygen? Science 2001; 292 (5516): 449–451
Acknowledgements
Niels B.J. Vollaard is grateful for a studentship provided by the Department of Biological Sciences, University of Essex, Essex, UK. The authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vollaard, N.B.J., Shearman, J.P. & Cooper, C.E. Exercise-Induced Oxidative Stress. Sports Med 35, 1045–1062 (2005). https://doi.org/10.2165/00007256-200535120-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00007256-200535120-00004