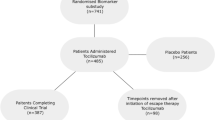
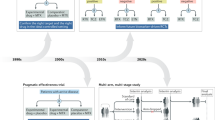
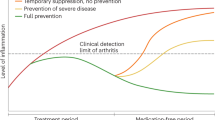
Abstract
Rheumatoid arthritis (RA) is a chronic, heterogeneous disease, for which there has traditionally been few reliable prognostic indicators or simple clinical outcome measures with which to adequately assess disease status or clinical improvement. These challenges have hindered the assessment of new therapeutic agents. Investigators in rheumatology have, therefore, attempted over the past two decades to develop new approaches and scoring systems to facilitate the study and development of therapies. As a result of these efforts, the efficacy of almost all new agents tested in clinical trials are now assessed with widely accepted scoring systems that measure improvement in three important areas. First, changes in patient signs and symptoms are measured via composite indices of both clinical and laboratory parameters. Second, the progression of structural damage is assessed by radiographic imaging. Finally, long-term improvement in patient function is measured via patient-reported outcomes. These changes to clinical trial design have been adopted by health authorities around the world. Further challenges, however, must be overcome in order for progress to continue.
Key Points
-
Three parameters are critical to the study of new investigational agents for rheumatoid arthritis (RA): clinical outcomes, radiographic outcomes and patient function
-
Two composite clinical outcome scoring systems—the American College of Rheumatology index and the Disease Activity Score—are widely used by rheumatologists in the study of RA
-
Several radiographic outcome measures are used in RA studies, including the Sharp and Larsen scores and their modifications, while patient function is assessed by the Health Assessment Questionnaire and Short Form-36
-
Careful selection of study participants, accurate study of dose effects, use of adequate comparators and long-term safety monitoring are all important features in the study of new investigational agents for RA
-
Challenges for the future include the need for new definitions for clinical and radiographic response, data on acceptable timing and duration of therapy, more-efficient trial designs, more studies of head-to-head comparisons and combination therapies, and increased emphasis on drug safety
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wolfe F (1996) The natural history of rheumatoid arthritis. J Rheumatol Suppl 44: 13–22
Rojkovich B and Poór G (2002) Prognostic factors in rheumatoid arthritis [Hungarian]. Orv Hetil 143: 2019–2026
FDA (1997) Guidance for industry: clinical development programs for drugs, devices and biological products for the treatment of rheumatoid arthritis (RA) [http://www.fda.gov/cber/gdlns/rheumcln.pdf] (accessed 15 October 2008)
Boers M et al. (1994) World Health Organization and International League of Associations for Rheumatology core endpoints for symptom modifying antirheumatic drugs in rheumatoid arthritis clinical trials. J Rheumatol Suppl 41: 86–89
Boers M et al. (1995) Assessing the activity of rheumatoid arthritis. Baillieres Clin Rheumatol 9: 305–317
Felson DT et al. (1995) American College of Rheumatology. Preliminary definition of improvement in rheumatoid arthritis. Arthritis Rheum 38: 727–735
Ranganath VK et al. (2006) ACR remission criteria and response criteria. Clin Exp Rheumatol 24 (6 Suppl 43): S14–S21
van Gestel AM et al. (1996) Development and validation of the European League Against Rheumatism response criteria for rheumatoid arthritis. Comparison with the preliminary American College of Rheumatology and the World Health Organization/International League Against Rheumatism Criteria. Arthritis Rheum 39: 34–40
The European Agency for the Evaluation of Medicinal Products (online 17 December 2003) Points to Consider on Clinical Investigation of Medicinal Products other than NSAIDs for Treatment of Rheumatoid Arthritis. [http://www.tga.gov.au/DOCS/pdf/euguide/ewp/055695enrev1.pdf] (accessed 15 Oct 2008)
Kremer JM (2007) Rheumatoid arthritis: a rapidly evolving treatment paradigm. Manag Care Interface 20: 17–24
Harth M and J Pope (2004) The measure of our measures. Rheumatology (Oxford) 43: 1465–1467
van der Heijde DM (2000) Radiographic imaging: the 'gold standard' for assessment of disease progression in rheumatoid arthritis. Rheumatology (Oxford) 39 (Suppl 1): 9–16
Boini S and Guillemin F (2001) Radiographic scoring methods as outcome measures in rheumatoid arthritis: properties and advantages. Ann Rheum Dis 60: 817–827
Fries JF et al. (1986) Assessment of radiologic progression in rheumatoid arthritis. A randomized, controlled trial. Arthritis Rheum 29: 1–9
Sharp JT et al. (1985). How many joints in the hands and wrists should be included in a score of radiologic abnormalities used to assess rheumatoid arthritis. Arthritis Rheum 28: 1326–1335
Larsen AK et al. (1977) Radiographic evaluation of rheumatoid arthritis and related conditions by standard reference films. Acta Radiol Diagn (Stockh) 18: 481–491
Van der Heijde DM et al. (1989) Effects of hydroxychloroquine and sulfasalazine on progression of joint damage in rheumatoid arthritis. Lancet 1: 1036–1038
Van der Heijde DM et al. (1992) Biannual radiographic assessments of hands and feet in a three year prospective follow-up of patients with early rheumatoid arthritis. Arthritis Rheum 35: 26–34
Van der Heijde DM (1999) How to read radiographs according to the Sharp/van der Heijde method. J Rheum 27: 261–263
Genant HK et al. (1998) Assessment of rheumatoid arthritis using a modified scoring method on digitized and original radiographs. Arthritis Rheum 41: 1583–1590
Pincus T et al. (2002) Evidence from clinical trials and long-term observational studies that disease-modifying anti-rheumatic drugs slow radiographic progression in rheumatoid arthritis: updating a 1983 review. Rheumatology 41: 1346–1356
Fries JF et al. (1980) Measurement of patient outcome in arthritis. Arthritis Rheum 23: 137–145
Leung YY et al. (2008) Comparison of 4 functional indexes in psoriatic arthritis with axial or peripheral disease subgroups using rasch analyses. J Rheum 35: 1613–1621
Thumboo J (2003) Measuring functional status in patients with systemic lupus erythematosus. APLAR J of Rheum 6: 184–187
Del Rosso A (2004) Health-related quality of life in systemic sclerosis as measured by short form 36: Relationship with clinical and biological markers. Arthritis Rheum 51: 475–481
Pollard LE et al. (2005) The consequences of rheumatoid arthritis: quality of life measures in the individual patient. Clin Exp Rheumatol 23 (5 Suppl 39): S43–S52
Bruce B and Fries JF (2005) The Health Assessment Questionnaire (HAQ). Clin Exp Rheumatol 23 (5 Suppl 39): S14–S18
Tugwell et al. (2008) Generic quality of life assessment in rheumatoid arthritis. Am J Manag Care 14: 234
Askling J and Bongartz T (2008) Malignancy and biologic therapy in rheumatoid arthritis. Curr Opin Rheumatol 20: 334–339
Taylor PC (2003) Anti-TNF alpha therapy for rheumatoid arthritis: an update. Intern Med 42: 15–20
Svenson M P et al. (2007) Monitoring patients treated with anti-TNF-alpha biopharmaceuticals: assessing serum infliximab and anti-infliximab antibodies. Rheumatology 46: 1828–1834
Olsen DP (2000) Equipoise: an appropriate standard for ethical review of nursing research. J Adv Nurs 31: 267–273
van der Heijde DM et al. (1993) Development of a disease activity score based on judgment in clinical practice by rheumatologists. J Rheumatol 20: 579–581
Ware JE Jr and Sherbourne CD (1992) The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 30: 473–483
Pincus T et al. (2004) Relative versus absolute goals of therapies for RA: ACR 20 or ACR 50 responses versus target values for “near remission” of DAS or single measures. Clin Exp Rheumatol 22 (5 Suppl 35): S50–S56
Sesin CA and Bingham CO III (2005) Remission in rheumatoid arthritis: wishful thinking or clinical reality. Semin Arthritis Rheum 35: 185–196
O'Dell J (2005) Rheumatoid arthritis initial therapy: unanswered questions. J Rheumatol Suppl 72: 14–16
Boers M (2008) COBRA combination therapy in daily practice—getting back to the future. Rheumatology 47: 1–2
Tugwell P et al. (2007) Generic quality-of-life assessment in rheumatoid arthritis. Am J Manag Care 13 (Suppl 9): S224–S236
Strand V et al. (2004) Patient-reported outcomes better discriminate active treatment from placebo in randomized controlled trials in rheumatoid arthritis. Rheumatology 43: 640–647
Heller JE and Shadick NA (2007) Outcomes in rheumatoid arthritis: incorporating the patient perspective. Curr Opin Rheumatol 19: 101–105
Pincus T (2006) Advantages and limitations of quantitative measures to assess rheumatoid arthritis: joint counts, radiographs, laboratory tests, and patient. Bull NYU Hosp Jt Dis 64: 32–39
Rau R (2006) Is remission in rheumatoid arthritis associated with radiographic healing. Clin Exp Rheumatol 24 (6 Suppl 43): S41–S44
Rau R G et al. (2004) Healing of erosive changes in rheumatoid arthritis. Clin Exp Rheumatol 22 (5 Suppl 35): S44–S49
Zierhut ML et al. (2007) Kinetic modeling of contrast-enhanced MRI: an automated technique for assessing inflammation in the rheumatoid arthritis wrist. Ann Biomed Eng 35: 781–795
Chen TS et al. (2006) Magnetic resonance imaging is more sensitive than radiographs in detecting change in size of erosions in rheumatoid arthritis. J Rheumatol 33: 1957–1967
Chiodo GT et al. (2000) Placebo-controlled trials. West J Med 172: 271–273
Fries JF and Krishnan E (2004) Equipoise, design bias, and randomized controlled trials. Arthrtis Res Ther 6: R250–R255
Fleischmann RM (2006) Comparison of the efficacy of biologic therapy for rheumatoid arthritis: can the clinical trials be accurately compared. Rheum Dis Clin North Am 32 (Suppl 1): S21–S28
Glocker MO et al. (2006) Rheumatoid arthritis, a complex multifactorial disease: on the way toward individualized medicine. Med Res Rev 26: 63–87
Koch A (2006) Confirmatory clinical trials with an adaptive design. Biom J 48: 574–585
Acknowledgements
I would like to acknowledge Donald J Schwieterman MD for his guidance and generosity, both recently and over the years.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Schwieterman, W. Issues in the design of new clinical trials for rheumatoid arthritis therapeutics. Nat Rev Rheumatol 4, 641–648 (2008). https://doi.org/10.1038/ncprheum0948
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncprheum0948
This article is cited by
-
Creative trial design in RA: optimizing patient outcomes
Nature Reviews Rheumatology (2013)