Abstract
Purpose
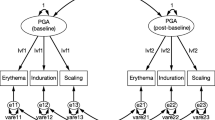
The Physician Global Assessment (PGA) is a key measure of psoriasis frequently used in clinical trials. A psychometric validation of a three-item (erythema, induration, and scaling) PGA scale was performed using Phase 2 data.
Methods
Confirmatory factor analysis (CFA) tested the PGA measurement model and appropriateness of equal weighting of the items. PGA test–retest reliability was assessed by estimating the intraclass correlation coefficient (ICC). Internal consistency reliability was gauged by calculating Cronbach’s coefficient α (CCα). Clinically important difference (CID) was defined using the repeated measures model to estimate the relationship between PGA and Patient Global Assessment (PtGA). Known-group, convergent, and divergent validity for the PGA were also assessed.
Results
197 patients with psoriasis were randomized to tofacitinib 2, 5, 15 mg twice daily, or placebo. CFA demonstrated that the PGA measurement model fitted the data using equal weighting of the PGA items. The PGA scale demonstrated good test–retest reliability (ICC > 0.7) and internal consistency reliability (CCα > 0.8). The CID for PGA was estimated at 0.52 (95 % confidence interval: 0.47, 0.56). A robust monotonic relationship between PGA and Psoriasis Area and Severity Index (PASI) data substantiated known-group validity. Relatively high correlations of PGA with PASI and PtGA data (all correlations >0.5 except at baseline) supported convergent validity; relatively low correlations of PGA with the Pain/Discomfort Assessment and the Ocular Comfort Index supported divergent validity.
Conclusions
The three-item PGA scale has sound psychometric properties with respect to reliability and validity, with equal weighting of the items being appropriate.




Similar content being viewed by others
References
Fredriksson, T., & Pettersson, U. (1978). Severe psoriasis—Oral therapy with a new retinoid. Dermatologica, 157, 238–244.
Feldman, S. R., & Krueger, G. G. (2005). Psoriasis assessment tools in clinical trials. Annals of the Rheumatic Diseases, 64(Suppl 2), ii65–ii68.
European Medicines Agency. (2004). Guideline on clinical investigation of medicinal products indicated for the treatment of psoriasis.
Ashcroft, D. M., Wan Po, A. L., Williams, H. C., & Griffiths, C. E. (1999). Clinical measures of disease severity and outcome in psoriasis: A critical appraisal of their quality. British Journal of Dermatology, 141, 185–191.
Langley, R. G., & Ellis, C. N. (2004). Evaluating psoriasis with psoriasis area and severity index, psoriasis global assessment, and lattice system physician’s global assessment. Journal of the American Academy of Dermatology, 51, 563–569.
Berth-Jones, J., Grotzinger, K., Rainville, C., et al. (2006). A study examining inter- and intrarater reliability of three scales for measuring severity of psoriasis: Psoriasis area and severity index, physician’s global assessment and lattice system physician’s global assessment. British Journal of Dermatology, 155, 707–713.
Spuls, P. I., Lecluse, L. L. A., Poulsen, M.-L., Bos, J. D., Stern, R. S., & Nijsten, T. (2010). How good are clinical severity and outcome measures for psoriasis? Quantitative evaluation in a systematic review. Journal of Investigative Dermatology, 130, 933–943.
Naldi, L. (2010). Scoring and monitoring the severity of psoriasis. What is the preferred method? What is the ideal method? Is PASI passé? facts and controversies. Clinics in Dermatology, 28(1), 67–72.
Naldi, L., Svensson, A., Diepgen, T., et al. (2003). Randomized clinical trials for psoriasis 1977–2000: The EDEN survey. Journal of Investigative Dermatology, 120, 738–741.
Robinson, A., Kardos, M., & Kimball, A. B. (2012). Physician global assessment and psoriasis area and severity index: Why do both? A systematic analysis of randomized controlled trials of biologics agents for moderate to severe plaque psoriasis. Journal of the American Academy of Dermatology, 66, 369–375.
Puzenat, E., Bronsard, V., Prey, S., et al. (2010). What are the best outcome measures for assessing plaque psoriasis severity? A systematic review of the literature. Journal of the European Academy of Dermatology and Venereology, 24(Suppl 2), 10–16.
Stern, R. S. (2010). Poor metrics and lost opportunity. Journal of the American Academy of Dermatology, 63, 718–719.
Sloan, J. A., Aaronson, N., Cappelleri, J. C., et al. (2002). Assessing the clinical significance of single items relative to summated scores. Mayo Clinic Proceedings, 77, 479–487.
Papp, K., Menter, A., Strober, B., et al. (2012). Efficacy and safety of tofacitinib, an oral JAK inhibitor, in the treatment of psoriasis: A Phase 2b randomised placebo-controlled dose-ranging study. British Journal of Dermatology, 167, 668–677.
Garduno, J., Bhosle, M. J., Balkrishnan, R., & Feldman, S. R. (2007). Measures used in specifying psoriasis lesion(s), global disease and quality of life: A systematic review. Journal of Dermatological Treatment, 18, 223–242.
Karabulut, A. A., Yalvac, I. S., Vahaboglu, H., et al. (1999). Conjunctival impression cytology and tear-film changes in patients with psoriasis. Cornea, 18, 544–548.
Johnson, M. E., & Murphy, P. J. (2007). Measurement of ocular surface irritation on a linear interval scale with the ocular comfort index. Investigative Ophthalmology & Visual Science, 48, 4451–4458.
Bentler, P. M. (1990). Comparative fit indexes in structural models. Psychological Bulletin, 107, 238–246.
Brown, T. A. (2006). Confirmatory factor analysis for applied research. New York, NY: The Guilford Press.
Hatcher, L. (1994). A step-by-step approach to using the SAS ® system for factor analysis and structural equation modeling. Cary, Nc: SAS Institute Inc.
Kline, R. (2011). Principles and practice of structural equation modeling (3rd ed.). New York, NY: The Guilford Press.
Fayers, F. M., & Machin, D. (2007). Quality of life: The assessment, analysis, and interpretation of patient-reported outcomes (2nd ed.). Chichester, England: Wiley.
Cappelleri, J. C., & Bushmakin, A. G. (2013). Interpretation of patient-reported outcomes. Statistical Methods in Medical Research (in press).
King, M. T. (2011). A point of minimal important difference (MID): A critique of terminology and methods. Expert Review of Pharmacoeconomics Outcomes Research, 11, 171–184.
Mulhall, J. O., Goldstein, I., Bushmakin, A., et al. (2007). Validation of the erectile hardness score. The Journal of Sexual Medicine, 4, 1626–1634.
Deyo, R. A., Inui, T. S., Leininger, J., et al. (1982). Physical and psychosocial function in rheumatoid arthritis: Clinical use of a self-administered health status instrument. Archives of Internal Medicine, 142, 879–882.
Cohen, J. (1988). Statistical power analysis for the behavioral sciences (2nd ed.). Hillsdale, NJ: Lawrence Erlbaum Associates.
Stevens, J. (2002). Applied multivariate statistics for the social sciences (4th ed.). Mahwah, NJ: Lawrence Erlbaum Associates.
Copay, A. G., Subach, B. R., Glassman, S. D., et al. (2007). Understanding the minimum clinically important difference: A review of concepts and methods. The Spine Journal, 7, 541–546.
Acknowledgments
Editorial support was provided by Jonny Miller, PhD, and Anne Marie Reid, PhD, of Complete Medical Communications and was funded by Pfizer Inc. The authors would like to thank the A3921047 investigators, staff and patients, and the A3921047 study team.
Conflict of interest
The study was funded by Pfizer Inc. Dr Cappelleri, Mr Bushmakin, and Dr Mamolo are employees of Pfizer Inc. Ms Harness was a full-time employee of Pfizer Inc during the conduct and reporting of the study and now works at Novartis Pharma AG, Basel, Switzerland. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cappelleri, J.C., Bushmakin, A.G., Harness, J. et al. Psychometric validation of the physician global assessment scale for assessing severity of psoriasis disease activity. Qual Life Res 22, 2489–2499 (2013). https://doi.org/10.1007/s11136-013-0384-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-013-0384-y