Abstract
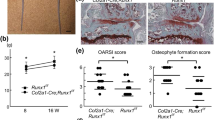
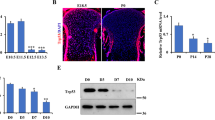
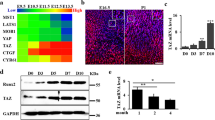
Endochondral ossification is an essential process not only for physiological skeletal development and growth, but also for pathological disorders. We recently identified a novel cartilage-specific molecule, carminerin (also known as cystatin 10 and encoded by Cst10), which is upregulated in synchrony with cartilage maturation and stimulates the later differentiation of cultured chondrocytes1. Although carminerin-deficient (Cst10−/−) mice developed and grew normally, they had a microscopic decrease in the calcification of hypertrophic chondrocytes at the growth plate. When we created experimental models of pathological endochondral ossification, we observed suppression of chondrocyte calcification during formation of osteoarthritic osteophytes, age-related ectopic ossification and healing of bone fractures in Cst10−/− mice. Cultured Cst10−/− chondrocytes showed a reduction in calcification with activation of an SRY site in the promoter of the gene encoding nucleotide pyrophosphatase phosphodiesterase 1 (NPP1, encoded by Enpp1). Functional NPP1 is required for carminerin deficiency to suppress the pathological endochondral ossifications listed above. Carminerin is the first cartilage-specific protein that contributes to chondrocyte calcification during endochondral ossification under physiological and pathological conditions through the transcriptional inhibition of NPP1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Koshizuka, Y. et al. Cystatin 10, a novel chondrocyte-specific protein, may promote the last steps of the chondrocyte differentiation pathway. J. Biol. Chem. 278, 48259–48266 (2003).
Kamekura, S. et al. Osteoarthritis development in novel experimental mouse models induced by knee joint instability. Osteoarthritis Cartilage 13, 632–641 (2005).
Mankin, H.J., Johnson, M.E. & Lippiello, L. Biochemical and metabolic abnormalities in articular cartilage from osteoarthritic human hips. III. Distribution and metabolism of amino sugar-containing macromolecules. J. Bone Joint Surg. Am. 63, 131–139 (1981).
Shimoaka, T. et al. Impairment of bone healing by insulin receptor substrate-1 deficiency. J. Biol. Chem. 279, 15314–15322 (2004).
Chikuda, H. et al. Cyclic GMP-dependent protein kinase II is a molecular switch from proliferation to hypertrophic differentiation of chondrocytes. Genes Dev. 18, 2418–2429 (2004).
Wolbach, S.B. Vitamin-A deficiency and excess in relation to skeletal growth. J. Bone Joint Surg. 29, 171–192 (1947).
Terkeltaub, R.A. Inorganic pyrophosphate generation and disposition in pathophysiology. Am. J. Physiol. Cell Physiol. 281, C1–C11 (2001).
Bollen, M., Gijsbers, R., Ceulemans, H., Stalmans, W. & Stefan, C. Nucleotide pyrophosphatases/phosphodiesterases on the move. Crit. Rev. Biochem. Mol. Biol. 35, 393–432 (2000).
Balcerzak, M. et al. The roles of annexins and alkaline phosphatase in mineralization process. Acta Biochim. Pol. 50, 1019–1038 (2003).
Ryan, L.M. The ank gene story. Arthritis Res. 3, 77–79 (2001).
de Crombrugghe, B., Lefebvre, V. & Nakashima, K. Regulatory mechanisms in the pathways of cartilage and bone formation. Curr. Opin. Cell Biol. 13, 721–727 (2001).
Akiyama, H., Chaboissier, M.C., Martin, J.F., Schedl, A. & de Crombrugghe, B. The transcription factor Sox9 has essential roles in successive steps of the chondrocyte differentiation pathway and is required for expression of Sox5 and Sox6. Genes Dev. 16, 2813–2828 (2002).
Lotz, M. et al. Interleukin 1 beta suppresses transforming growth factor-induced inorganic pyrophosphate (PPi) production and expression of the PPi-generating enzyme PC-1 in human chondrocytes. Proc. Natl. Acad. Sci. USA 92, 10364–10368 (1995).
Solan, J.L., Deftos, L.J., Goding, J.W. & Terkeltaub, R.A. Expression of the nucleoside triphosphate pyrophosphohydrolase PC-1 is induced by basic fibroblast growth factor (bFGF) and modulated by activation of the protein kinase A and C pathways in osteoblast-like osteosarcoma cells. J. Bone Miner. Res. 11, 183–192 (1996).
Oyajobi, B.O., Caswell, A.M. & Russell, R.G. Transforming growth factor beta increases ecto-nucleoside triphosphate pyrophosphatase activity of human bone-derived cells. J. Bone Miner. Res. 9, 99–109 (1994).
Okawa, A. et al. Mutation in Npps in a mouse model of ossification of the posterior longitudinal ligament of the spine. Nat. Genet. 19, 271–273 (1998).
Goldfine, I.D., Maddux, B.A., Youngren, J.F., Trischitta, V. & Frittitta, L. Role of PC-1 in the etiology of insulin resistance. Ann. NY Acad. Sci. 892, 204–222 (1999).
Shukunami, C. et al. Chondrogenic differentiation of clonal mouse embryonic cell line ATDC5 in vitro: differentiation-dependent gene expression of parathyroid hormone (PTH)/PTH-related peptide receptor. J. Cell Biol. 133, 457–468 (1996).
Laroche, M. Phosphate, the renal tubule, and the musculoskeletal system. Joint Bone Spine 68, 211–215 (2001).
Jono, S. et al. Phosphate regulation of vascular smooth muscle cell calcification. Circ. Res. 87, E10–E17 (2000).
Nakamichi, Y. et al. Chondromodulin I is a bone remodeling factor. Mol. Cell. Biol. 23, 636–644 (2003).
Parfitt, A.M. et al. Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner. Res. 2, 595–610 (1987).
Acknowledgements
This study was supported by a Grant-in-aid for Scientific Research from the Japanese Ministry of Education, Culture, Sports, Science, and Technology (#14370454), and by the Investigation Committee on the Ossification of Spinal Ligaments, Japanese Ministry of Public Health and Welfare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Radiological and histological findings of tibias and vertebrae under physiological conditions. (PDF 587 kb)
Supplementary Fig. 2
Radiological and histological findings of age-related ectopic ossification. (PDF 469 kb)
Supplementary Fig. 3
Ex vivo cultures of growth plate chondrocytes and calvarial osteoblasts. (PDF 989 kb)
Supplementary Fig. 4
Effects of putative cytokines on the carminerin signaling to inhibit NPP1. (PDF 190 kb)
Supplementary Fig. 5
Osteoarthritic osteophyte formation, age-related ectopic ossification, and high phosphate-induced auricular ossification in mice of four genotypes. (PDF 567 kb)
Supplementary Table 1
Dissociation constants of complexes between cystatins and cysteine proteinases. (PDF 133 kb)
Rights and permissions
About this article
Cite this article
Yamada, T., Kawano, H., Koshizuka, Y. et al. Carminerin contributes to chondrocyte calcification during endochondral ossification. Nat Med 12, 665–670 (2006). https://doi.org/10.1038/nm1409
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1409
This article is cited by
-
CCL21/CCR7 axis regulating juvenile cartilage repair can enhance cartilage healing in adults
Scientific Reports (2019)
-
Association between metabolic syndrome and knee osteoarthritis: a cross-sectional study
BMC Musculoskeletal Disorders (2017)
-
Does osteophytosis at the knee predict health-related quality of life decline? A 3-year follow-up of the ROAD study
Clinical Rheumatology (2015)
-
Association of dietary intake with joint space narrowing and osteophytosis at the knee in Japanese men and women: the ROAD study
Modern Rheumatology (2013)
-
The Samd9L Gene: Transcriptional Regulation and Tissue-Specific Expression in Mouse Development
Journal of Investigative Dermatology (2011)