Abstract
The primary objective of this investigation was to assess the relationships between clinical characteristics, lung involvement, and frequency of pulmonary involvement in rheumatoid arthritis (RA). Using high-resolution computed tomography (HRCT) and pulmonary function tests (PFT), we prospectively evaluated 52 patients with RA (eight males and 44 females, mean age 53.6 years). The HRCT was abnormal in 35 patients (67.3%), the most frequent abnormalities being reticulonodular patterns, which were found in 22 patients (62.9%), ground-glass attenuation (20%), and bronchiectasis (17%). In this group of patients, PFT results were normal in 13 patients (37%). Titers of rheumatoid factor and erythrocyte sedimentation rate were significantly higher in abnormal HRCT presence. Higher Larsen’s score, advanced age, and severe disease were significant risk factors for lung involvement (p<0.001, p<0.01, and p<0.01, respectively) and are suggested by our data to be statistically significant predictors of lung involvement in RA.
Similar content being viewed by others
Introduction
Pulmonary involvement is a frequent extra-articular manifestation of rheumatoid arthritis (RA) and might be the second most frequent cause of death, after infection. Interstitial lung disease (ILD) and subclinical alveolitis have been found in up to 40% of RA patients. In general, ILD is seen more frequently in men than women, in the presence of high rheumatoid factor (RF) titers, and in the setting of more severe articular disease. It is often a progressive disease which may result in disabling symptoms and respiratory failure [1, 2].
Radiographically, the pulmonary changes seen in RA are indistinguishable from those seen with idiopathic pulmonary fibrosis or ILD associated with other connective tissue disease. Although ILD is a well-known manifestation of RA, small-airway involvement may be the most common form of RA lung involvement. Apart from pulmonary function tests (PFTs), other noninvasive tools such as high-resolution computed tomography (HCRT) of the lungs are more sensitive in the assessment of rheumatoid lung disease [3, 4, 5, 6]. This prospective study aimed to assess by HCRT and PFT the frequency and predictive factors of pulmonary involvement in unselected RA patients.
Patients and methods
The 54 patients were diagnosed with RA according to the 1987 American Rheumatism Association criteria. Those with histories of chronic obstructive lung disease, pleuropulmonary tuberculosis, thoracic radiation for breast carcinoma, or pleural effusion were excluded.
We noted the duration of RA, extra-articular complications, duration of early-morning joint stiffness, current and previous disease-modifying antirheumatic drugs (DMARDs), and corticosteroid use. Clinical evaluation of articular disease was based on the Ritchie articular index (range 0–78, in which 78 represents maximal pressure-induced pain in all joints), number of swollen joints, patient’s global assessment of disease severity, pain on a 10-cm visual analog scale, and morning stiffness duration. Each patient filled in the Arthritis Impact Measurement Scale 2 short form (AIMS2-SF) to assess quality of life and thus provide a measure of long-term disease severity.
Patients were asked about cough, dyspnea, sputum production, chest pain, weight loss, alcohol habits, previous chest disease, and risk factors for respiratory disease such as smoking, drugs, domestic pets, and occupation. Current smokers were those who had smoked more than five cigarettes a day during the previous 6 months; nonsmokers had smoked less than 20 packets of cigarettes during their lifetimes.
All patients had venous blood taken for full blood count, erythrocyte sedimentation rate (ESR), renal and liver function, C-reactive protein (CRP), and plasma proteins. Immunological investigations included RF, antinuclear antibodies (ANA), and human leukocyte antigen (HLA)-DR1 and -DR4 genotypes. The latter genotypes were determined by polymerase chain reaction-restriction fragment length polymorphism. All patients underwent chest radiography, HRCT, and full PFTs. After being informed about the objective, all gave their informed consent. Pulmonary function testing and HRCT scans were always performed less than a week apart. Radiographs of the hand were performed to calculate Larsen’s scores. Carbon monoxide diffusion capacity was not assessed in our patients because of technical limitations. The patients were examined for the presence of extra-articular manifestations (e.g., vasculitis, nodules, Sjögren’s syndrome). In the absence of clinical information, the HRCT examinations were interpreted by two experienced radiologists and the decision was obtained by consensus.
Pulmonary function testing
Lung function was measured by a standard protocol and included spirometry. The following PFTs were performed: forced expiratory volume in 1 s (FEV1), forced vital capacity (FVC), peak expiratory flow (PEF), FEV1/FVC, and forced expiratory flow between 25% and 75% of vital capacity (FEF 25–75). The results were expressed in percentages. Pulmonary function test abnormalities were categorized as abnormal if values were <80% of the predicted values adjusted for age, sex, and height.
According to the PFTs results, three groups were defined: (1) airway obstruction defined by a low FEV1/FVC ratio, low PEF value, or low FEF25–75 value in the absence of reduced FEV1/FVC ratio; (2) restrictive disorder defined by a decrease in FVC and FEV1 with a normal FEV1/FVC ratio; and (3) a combined pattern defined as coexistence of airway obstruction and restrictive syndrome.
High-resolution computed tomography of the lungs
The HRCT scans in the workup studies were performed using a spiral CT scanner (Xpress/GX, TSX-002a) (Toshiba, Tocigi-Ken, Japan). The technical scan parameters were 130 kV and 200 mAs. In all cases, HRCT images were obtained at inspiration using a slice thickness of 2 mm at 10-mm intervals and reconstructed using a high spatial frequency algorithm. The images were displayed at window and level settings which optimized the visualization of lung parenchyma (level −600 to −700 HU, width 1000–1600 HU). The HRCT examinations were analyzed by two radiologists (C.C. and M.D.), and a consensus opinion was obtained. Parenchymal and alveolar features were examined.
Interpretation of the tomographic scans
A grading system based on the observations of Müller et al. [7] was used to categorize the relative extents of the reticular and ground-glass patterns as follows: grade 1: ground-glass opacities predominate; grade 2: reticular pattern predominates; and grade 3: reticulonodular (fibrotic) lesion pattern predominates. Bronchiectasis was defined as bronchial dilatation.
Statistical analysis
Student’s t-test was used to compare normally distributed quantitative data, and the Mann-Whitney U test was used to compare quantitative data not distributed normally. The chi-squared test with Yates correction was used to compare frequencies. Potentially significant parameters were tested for possible interrelationship by multiple logistic regression analysis. Correlations between variables were assessed with Pearson’s and Spearman’s rank correlation tests. Possible relationships between PFTs with HRCT findings were tested using analysis of variance. Probability values of <0.05 were considered to be statistically significant.
Results
Fifty-four patients with RA according to the 1987 American College of Rheumatology criteria and aged 53.63±11.19 years (range 27–76, 46 women and eight men) were included in the study. Pulmonary function tests could not be evaluated in two of the women, who were excluded from all analyses. Thus, 52 patients were included in data analysis. The mean duration of articular disease was 8.37±8.17 years at the time of pulmonary evaluation. The mean AIMS2-SF score was 76.44±9.49. Mean ESR was 51.21±29.39 mm/h. Thirty-five patients were positive for RF, and 12 were ANA-positive. Basic clinical and demographic data of the patients are summarized in Table 1. Human leukocyte antigen DR4 was positive in 21 patients, and HLA-DR1 was positive in nine. However, both HLA-DR1 and HLA-DR4 were positive in two patients.
Thirty-three patients (63%) were taking oral steroids and 38 (73%) were taking DMARDs at the time of assessment. Four patients were not receiving DMARDs. Thirty-five (67%) were taking methotrexate (MTX), 32 (61%) sulfasalazine, 16 hydroxychloroquine (31%), two cyclosporin A, four infliximab, and one leflunomid. However, drug history was difficult to analyze, since the patients had taken a mean of 4±1.3 second-line drugs for RA at the time of assessment.
Thirty-one patients had no respiratory symptoms. Among those with respiratory symptoms, dyspnea was the most common, followed by cough and sputum. Eleven patients complained of the following respiratory symptoms: dyspnea (n=8, all nonsmokers), and chronic cough (n=6, including two smokers), which was associated with chronic purulent sputum in three smokers and one nonsmoker. One patient was suffering from Sjögren’s syndrome. Subcutaneous rheumatoid nodules were noted in two cases.
Pulmonary function tests
Our study subjects had mean PFTs values within the normal range. Airway obstruction was noted in 12 patients, pure restrictive defect in three, and a combination of both in 14 cases. The PFTs values were normal in 13 of the 35 patients with lung involvement confirmed by HRCT. Two of 12 patients with airway obstruction were smokers.
Seventy-five percent of the patients participating in this study were lifelong nonsmokers, and 13 were current or former smokers. Among the 13 smokers with RA, PFTs showed combined impairment in four and obstructive impairment in three. The results of the PFTs were not different between the 39 nonsmokers and former or current smokers (Table 2).
We found significant correlations between RF values and FEV1 and PEF (r=−0.27 and p<0.05, r=−0.39 and p<0.01, respectively). There was also a correlation between PEF and Larsen’s score (r=−0.33, p<0.05). The FVC, FEV1, and PEF values showed significant decreases with patient age (p<0.01, p<0.05, and p<0.05, respectively). Although PFTs values were generally lower in patients receiving MTX, this was not statistically significant.
High-resolution computed tomographic findings
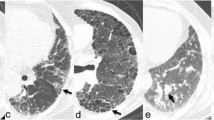
The HRCT scans were normal in 17 patients (32.6%) and abnormal in 35 (67.3%) (Table 3). Twenty-two had advanced disease with reticulonodular patterns (Fig. 1, Fig. 2), while seven also had ground-glass opacities. In three patients, these features overlapped. Bronchiectasis was found in six and associated with a reticulonodular pattern in three. Sixty-five percent of patients with lung involvement by HRCT had abnormal chest radiographs.
Posteroanterior chest view of the same patient as in Fig. 1 demonstrates a bilateral reticulonodular pattern
Among the patients with normal HRCT scans, PFTs were normal in ten, whereas obstructive patterns were found in seven. In the group with normal PFT (n=23), HRCT scans were normal in ten (43.5%) and showed various abnormalities in the remaining 13. Only seven of 35 patients with abnormal HRCT findings were smokers.
Relationship between clinical data and pulmonary function tests or high-resolution computed tomography findings
Rheumatoid arthritis showed the expected association with HLA-DR4 (40%), but patients with RA and coexistent pulmonary fibrosis were less likely to be DR4-positive (31%). Eleven patients with abnormal HRCT findings had HLA-DR4, and five had HLA-DR1. The HLA-DR genotype was not different between patients with or without HRCT findings. There were no significant differences in the frequencies of DR4 or DR1 genotypes between the different HRCT groups. Although there was a slight increase in DR4 and DR1 in RA combined with airway disease, this was not significant, either. There were no significant differences between PFTs of the HLA-DR4-positive and HLA-DR1-positive groups.
Four patients among six with HRCT identification of diffuse bronchiectasis did not complain of chronic repeated sputum and cough. Conversely, only two of the eight patients with these symptoms had normal HRCT scans. Two patients with bronchiectasis had HLA-DR4; only one had HLA-DR1. Rheumatoid factor was positive in all patients with bronchiectasis. Among the six patients with bronchiectasis, PFTs demonstrated obstruction patterns in two patients and combined patterns in the remaining four.
Pulmonoary function tests showed a significant difference between patients with bronchiectasis and those with normal HRCT findings. The mean values of FEV1 and PEF were significantly lower in patients with bronchiectasis than in the normal HRCT group (p<0.05) (Table 3). Abnormal HRCT findings were statistically significantly associated with Larsen’s radiological score, age, and patient global assessment of disease severity (odds ratio [OR] 331.37, 95% confidence interval [CI] 4.97–2275.3, p<0.001; OR=1.2395, 95% CI=1.03–1.52, p<0.01; and OR= 2.04, 95% CI=1.14–3.66, p<0.01, respectively).
High titers of RF were associated with lung involvement on HRCT. Statistically, titers of RF and ESR were significantly higher in patients with abnormal HRCT than those with normal HRCT. Eighty percent of patients with abnormal HRCT findings had RF positivity (n=28). The HRCT findings were unrelated to other rheumatological data such as CRP, ANA, AIMS2-SF, Ritchie articular index, pain score, and morning stiffness duration. Extra-articular manifestations showed no relationship with HRCT findings and were demonstrated by only three patients in our study.
The PFTs results did not correlate to disease duration or serological disease activity parameters including ESR, CRP, and pain score. No correlation was found between AIMS2-SF or Ritchie articular index and PFT variables. Four patients were treated after HRCT scans with regimens of prednisolone (minimum daily dose 10 mg), and a combination with azathioprine (100–150 mg daily) was given in two of 35 cases.
Discussion
We prospectively assessed the lungs of 52 patients with RA clinically, using chest radiography, pulmonary tests, and thoracic HRCT scanning. Prior investigations of the risk factors for lung involvement of RA have yielded disparate results. Traditional disease severity measures such as the presence of serum RF and subcutaneous nodules, antirheumatic therapies, immunogenetic disease severity markers such as HLA-DR4, and cigarette smoking have been identified by some authors as risk factors for lung involvement in RA [1, 5, 6, 8, 9]. However, the high prevalence of smokers in the previously published studies suggests that smoking might be the main cause of rheumatoid lung involvement. Conversely, cigarette smoking was not associated with physiological or HRCT abnormalities suggestive of RA-ILD in our study. This might be explained by the lower number particularly of current smokers (n=8). Saag et al. [10] reported that smoking was the most consistent independent risk factor predicting the development of ILD in RA. Krishman et al. [11] recently published that past smoking was strongly associated with development of RF-positive RA (OD 2.3, 95% CI 1.3–3.9) for men, but this was not the case for women.
Cortet et al. [12] observed large airway obstruction in 9% and small airway obstruction in 14% of patients with RA. Vergnenegre et al. [13] reported the ratio of airway obstruction in patients with RA to be 16% after the exclusion of smokers. Our study showed obstructive pulmonary involvement in 23% of RA patients. We observed that purely restrictive pulmonary function test results were rare, and this was interesting, but 48% of patients with abnormal HRCT findings had combined ventilatory impairment. These findings suggest the possible development of a restrictive defect over time.
Although smoking is known as a major risk factor for obstructive airway disease, the exact mechanism for the development of this obstruction in RA is not yet known. On the other hand, studies in nonsmoking RA patients show that smoking increases obstructive pulmonary involvement but does not play a big part in the process [12, 13]. Reduced respiratory muscle strength, aerobic capacity, and endurance may be due to pulmonary impairment.
Interstitial lung disease is a well-recognized manifestation of rheumatoid lung involvement. Four prospective studies reported an ILD prevalence of 19–44%. Also, the HRCT study by Fewins et al. [3] of patients with RA revealed a high prevalence of ILD (44%). Our data confirm a rather high prevalence of lung involvement in patients with RA (67.3%). These results are in agreement with those published by Cortet et al. [12]. They showed a high prevalence of lung involvement in patients with RA by HRCT (80.9%), a similar prevalence to that found by us. In our study, the most frequently encountered HRCT finding was reticulonodular pattern; the second most frequent was ground-glass pattern. Dawson et al. [14] reported that, in 82% of such patients, the RA-associated fibrosing alveolitis was predominantly of a reticular pattern. This prevalence is closer to that found in our study.
We identified bronchiectasis in 17% of patients with abnormal HRCT. Bronchiectasis frequency ranged from 20% to 30%, according to previous reports [14, 15]. Our results are in accordance with those of Demir et al. [15], who reported a 26% incidence of bronchiectasis and 68% abnormal patterns in their RA patients on HRCT. Some authors believed that bronchiectasis might precede RA by many years and that chronic suppuration in the lung may lead to antigenic stimulation in susceptible individuals, thus causing RA. However, it is difficult to explain whether bronchiectasis preceded RA in our patients, since four of six patients with bronchiectasis had no respiratory symptoms.
We did not observe a statistically significant association between PFTs results and the presence of lung involvement by HRCT. In contrast to our findings, McDonagh et al. [16] found that ground-glass attenuation was associated with a significant decrease in FEV1. These differences may be partly explained by the different smoking habits of the study groups. Cortet et al. [12] found that ground-glass attenuation was associated with a significant decrease in FVC. They also found low values of FEV1, FVC, and FEF25–75 in patients with bronchiectasis. In our study, severity of lung involvement was associated with the development of deterioration in gas exchange. Both FEV1 and FVC showed declines in patients with bronchiectasis and reticulonodular patterns. These changes were not statistically significant. Moreover, patients with bronchiectasis had significantly lower PEF and FVC values than those with normal HRCT findings. These results indicate that HRCT scanning detects lung involvement at a much earlier stage, before significant loss of lung volume and pulmonary compliance.
Gabbay et al. [4] prospectively studied 36 patients with RA of less than 2-year duration for ILD. They noted an association between male sex and clinically significant lung disease in their patients with early-onset RA, a different population from ours. Since the number of male patients was also significantly lower than that of females, we did not observe an association between male gender and lung involvement. We also found no relation between the presence of lung involvement and previously described predisposing factors such as nodular disease, extra-articular disease, and longer disease duration. Biederer et al. [17] recently reported that the graduation of lung parenchyma lesion patterns did not correlate with the duration of disease, either, although reticular patterns were found to predominate in patients with disease of long duration, whereas a predominance of ground-glass patterns was found more frequently in patients with shorter disease course. The high prevalence of reticular lesion patterns in patients with longer disease course suggests the development of fibrosis as a sequel of ground-glass infiltration.
Perez et al. [18] could demonstrate no relationship between RA disease parameters and PFTs or HRCT findings. Their findings contrast to those of Vergnenegre and associates, who reported a significant relationship between FEF 25–75 and duration of articular disease and between FEV1/FVC and the Ritchie index. In the present study, RF and ESR values were significantly higher in the presence lung involvement than in its absence. Nevertheless, RF and ESR were not among the risk factors for lung involvement. To identify risk factors when a stepwise strategy in a multiple logistic regression model was used, the high Larsen’s score, advanced age, and severity of disease in our study were found to be predictive of lung involvement. In contrast to our findings, Dawson et al. [14] found that the presence of fibrosing alveolitis did not relate with disease severity, either and they used Health Assessment questionnaire to assess the severity of disease.
A nonsignificant increase in HLA-DR4 was found in the combined group compared with other patients. Our study also suggests that the absence of an association between lung involvement and HLA-DR genotypes is in agreement with the results of Hillarby et al. [19] and Hassan et al. [20]. Rheumatoid arthritis with bronchiectasis has previously been said to be associated with HLA-DR1 [21]. Although we did not find any significant association of HLA-DR1 and bronchiectasis in our patients, this could reflect the relatively small number of bronchiectasis patients in our study.
Methotrexate, currently the most widely used second-line agent, was also the most widely used drug in our study. The study was not designed to investigate the incidence of pulmonary effects of MTX in patients with RA. Because of the possible influence of antirheumatic drugs on the lung, we analyzed the relationship between abnormal PFTs or HRCT scan results and current treatment with MTX. Despite the prevailing widespread use of MTX in RA, only a few studies have addressed the role of PFTs in predicting its pulmonary toxicity. Cotin et al. [22] data showed a slight decrease in FVC and FEV1 in the whole population of asymptomatic patients during MTX treatment. In previous studies that serially monitored PFTs in patients with RA on MTX, the authors concluded that, although there was deterioration of pulmonary function parameters, this did not reflect a clinically significant deterioration in RA patients treated with MTX [23, 24, 25]. Also, we found no significant difference in pulmonary function changes between the patients with and without MTX therapy.
Finally, second-line drugs and particularly MTX do not influence the results of PFT. There were no significant differences in pulmonary function or HRCT data in patients given different treatments. However, the complexity of previous drug histories precluded meaningful evaluation of the relationship between each single drug and pulmonary involvement.
Pulmonary involvement is an important part of the systemic affection of RA. Other systemic aspects of RA such as vascular disease or inflammatory activity may also influence lung disease. As with other connective tissue disease, it is commonly assumed that immune-mediated processes in RA are central to the inflammatory processes in the lung. Immune complex deposition in the lung interstitium and alveolar walls may contribute to alveolar macrophage activation. Cellular hypersensitivity mechanisms may also play a role in the pathogenesis. In early disease, these changes may appear as patchy alveolar filling infiltrates. Disease progression results in a more reticulonodular pattern [1, 2].
Although chest radiography is helpful in the assessment of ILD, it rarely allows a confident diagnosis [26]. A number of recent studies have suggested that HRCT of the chest is superior to chest radiography because the decreased superimposition of structures on HRCT images allows better assessment of the type, distribution, and severity of parenchymal abnormalities than is possible on radiography. Pulmonary function tests failed to predict HRCT scan findings in most of our patients. This is in keeping with previous observations. It appears that pulmonary function alone is insufficient for guiding diagnosis.
Our results also indicate that clinical complaints and PFT have low diagnostic accuracy in the early detection of pulmonary involvement in RA. In addition, serological findings, including titers of RF and ESR, seem to be of little value in clinical investigation of the pulmonary disease.
In summary, we found associations between severity of rheumatoid disease and the presence of lung involvement. Such involvement is an important part of the systemic affection of RA. Thus, it must always be investigated in erosive RA, even in the absence of respiratory symptoms. Our findings indicate that the severity of RA is an important predictor of HRCT abnormalities suggestive of lung involvement. We would recommend that, in the presence of severe RA such as that determined by high Larsen’s score, careful evaluation by HRCT scan should be requested in addition to clinical examination, chest radiography, and full PFT.
References
Tanoue LT (1998) Pulmonary manifestations of rheumatoid arthritis. Clin Chest Med 19:667–685
Du Bois RM, Wells AU (2000) Infiltrative and interstitial lung diseases. In: Murray JF, Nadel JA (eds) Textbook of respiratory medicine. Saunders, Philadelphia
Fewins HE, McGowan I, Whitehouse GH, Williams J, Mallya R (1991) High definition computed tomography in rheumatoid arthritis associated pulmonary disease. Br J Rheumatol 30:214–216
Gabbay E, Tarala R, Will R, Carroll G, Adler B, Cameron D, Lake FR (1997) Interstitial lung disease in recent onset rheumatoid arthritis. Am J Respir Crit Care Med 156:528–535
Mathieson JR, Mayo JR, Staples CA, Muller NL (1989) Chronic diffuse infiltrative lung disease: comparison of diagnostic accuracy of CT and chest radiography. Radiology 171:111–116
Wells AU, Rubens MB, Du Bois RM, Hansell DM (1993) Serial CT in fibrosing alveolitis: prognostic significance of the initial pattern. AJR Am J Roentgenol 161:1159–1616
Müller NL, Staples CA, Miller RR, Vedal S, Thurlbeck WM, Ostrow DN (1987) Disease activity in idiopathic pulmonary fibrosis: CT and pathological correlation. Radiology 165:731–734
Xaubet A, Agusti C, Luburich P, Roca J, Monton C, Ayuso MC, Barbera JA, Roisin RR (1998) Pulmonary function tests and CT scan in the management of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 158:431–436
Hakala M (1988) Poor prognosis in patients with rheumatoid arthritis hospitalized for interstitial lung fibrosis. Chest 93:114–118
Saag KG, Kolluri S, Koehnke RK, Georgou TA, Rachow JW, Hunninghake GW, Schwartz DA (1996) Rheumatoid arthritis lung disease. Determinant of radiographic and physiologic abnormalities. Arthritis Rheum 39:1711–1719
Krishnan E, Sokka T, Hannonen P (2003) Smoking-gender interaction and risk for rheumatoid arthritis. Arthritis Res Ther 5:158–162
Cortet B, Perez T, Roux N, Flipo RM, Duquesnoy B, Delcambre B, Jardin MR (1997) Pulmonary function tests and high resolution computed tomography of the lungs in patients with rheumatoid arthritis. Ann Rheum Dis 56:596–600
Vergnenegre A, Pugnere N, Antonini MT, Arnaud M, Melloni B, Treves R, Bonnaud F (1997) Airway obstruction and rheumatoid arthritis. Eur Respir J 10:1072–1078
Dawson JK, Fewins HE, Desmond J, Lynch MP, Graham DR (2001) Fibrosing alveolitis in patients with rheumatoid arthritis as assessed by high resolution computed tomography, chest radiography, and pulmonary function tests. Thorax 56:622–627
Demir R, Bodur H, Tokoğlu F, Olcay I, Uçan H, Borman P (1999). High resolution computed tomography of the lungs in patients with rheumatoid arthritis. Rheumatol Int 19:19–22
McDonagh, Greaves M, Wright AR, Heycock C, Owen JP, Kelly C (1994) High resolution computed tomography of the lungs in patients with rheumatoid arthritis and interstitial lung disease. Br J Rheumatol 33:118–122
Biederer J, Schnabel A, Muhle C, Gross WL, Heller M, Reuter M (2004) Correlation between HRCT findings, pulmonary function tests and bronchoalveolar lavage cytology in interstitial lung disease associated with rheumatoid arthritis. Eur Radiol 14:272–280
Perez T, Remy-Jarden M, Cortet B (1998) Airway involvement in rheumatoid arthritis. Am J Respir Crit Care Med 157:1658–1665
Hillarby MC, McMahon MJ, Grennan DM, Cooper RG, Clarkson RWE, Davies EJ, Sanders PA, Chattopadhyay C, Swinson D (1993) HLA associations in subjects with rheumatoid arthritis and bronchiectasis but not with other pulmonary complications of rheumatoid disease. Br J Rheumatol 32:794–797
Hassan WU, Keaney NP, Holland CD, Kelly CA (1995) High resolution computed tomography of the lung in lifelong nonsmoking patients with rheumatoid arthritis. Ann Rheum Dis 54:308–310
Chattopadhyay C, Shelton S, Swinson D et al. (1991) RA and bronchiectasis an immunogenetic study. Br J Rheumatol 28:34
Cottin V, Tebib J, Massonnet B, Souquet PJ, Bernard JP (1996) Pulmonary function in patients receiving long-term low-dose methotrexate. Chest 109:933–938
Dawson JK, Graham DR, Desmond J, Fewins HE, Lynch MP (2002) Investigation of the chronic pulmonary effects of low-dose oral methotrexate in patients with rheumatoid arthritis: a prospective study incorporating HRCT scanning and pulmonary function tests. Rheumatology (Oxford) 41:262–267
Beyeler C, Jordi B, Gerber NJ, Im Hof V (1996) Pulmonary function in rheumatoid arthritis treated with low-dose methotrexate: a longitudinal study. Br J Rheumatol 35:446–452
Khadadah ME, Jayakrishnan B, Gorair SA, Mutairi MA, Maradni NA, Onadeko B, Malaviya AN (2002) Effect of methotrexate on pulmonary function in patients with rheumatoid arthritis—a prospective study. Rheumatol Int 22:204–207
Perez PC, Perez AHT, Jurado MPR (1980) Pulmonary involvement in rheumatoid arthritis. JAMA 243:1715–1719
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bilgici, A., Ulusoy, H., Kuru, O. et al. Pulmonary involvement in rheumatoid arthritis. Rheumatol Int 25, 429–435 (2005). https://doi.org/10.1007/s00296-004-0472-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-004-0472-y